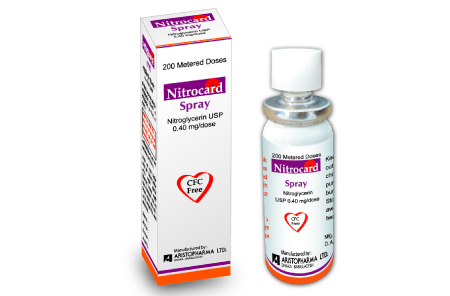
Nitrocard 1 Pcs
Alternative products
Nitroglycerin (Sublingual Spray)
Indications
Nitroglycerin sublingual spray is indicated for acute relief of an attack or prophylaxis of angina pectoris due to coronary artery disease.
Description
Nitroglycerin, an organic nitrate, is a vasodilator which has effects on both arteries and veins. This is a metered dose spray containing nitroglycerin. This product delivers nitroglycerin (400 mcg per spray, 200 metered sprays) in the form of spray droplets under the tongue.
Pharmacology
The principal pharmacological action of nitroglycerin is relaxation of vascular smooth muscle, producing a vasodilator effect on both peripheral arteries and veins with more prominent effects on the latter. Dilation of the post-capillary vessels, including large veins, promotes peripheral pooling of blood and decreases venous return to the heart, thereby reducing left ventricular enddiastolic pressure (pre-load). Arteriolar relaxation reduces systemic vascular resistance and arterial pressure (after-load).
Dosage & Administration
At the onset of an attack, 1 or 2 metered sprays should be administered under the tongue. No more than 3 metered sprays are recommended within a 15 minute period. If the chest pain persists, prompt medical attention is recommended. Nitroglycerin spray may be used prophylactically 5 to 10 minutes prior to engaging in activities which might precipitate an acute attack.
* চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Interaction
Use of alcohol with Nitroglycerin may produce severe hypotension and collapse. Oral Nitroglycerin may enhance the bioavailability of dihydroergotamine. Orthostatic hypotension may occur with the combined use of calcium channel blocker, phenothiazines and tricyclic antidepressants.
Contraindications
Hypersensitivity to nitrates or any constituents of the formulation. Hypotension, hypovolaemia, severe anaemia, cerebral haemorrhage and brain trauma, mitral stenosis and angina caused by hypertrophic obstructive cardiomyopathy. Concomitant administration of phosphodiesterase inhibitors used for the treatment of erectile dysfunction.
Side Effects
A number of nitrate related adverse effects may occur including headache, facial flushing, dizziness, nausea, vomiting, feelings of weakness, postural hypotension and reflex tachycardia .
Pregnancy & Lactation
Pregnancy Category C. There are no adequate and well controlled studies in pregnant women. Nitroglycerin should be given to pregnant women only if clearly needed. It is not known whether nitroglycerin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Nitroglycerin spray is administered to a nursing woman.
Precautions & Warnings
The use of Nitroglycerin during the early days of acute myocardial infarction requires particular attention to monitoring hemodynamics and clinical status. Nitroglycerin should be used with caution in patients with severely impaired renal or hepatic function, hypothyroidism, malnutrition or hypothermia.
Use in Special Populations
Safety and effectiveness of nitroglycerin in pediatric patients have not been established.
Overdose Effects
Nitrate overdosage may result in: severe hypotension, persistent throbbing headache, vertigo, palpitation, visual disturbance, flushing and perspiring skin (later becoming cold and cyanotic), nausea and vomiting (possibly with colic and even bloody diarrhea), syncope (especially in the upright posture), methemoglobinemia with cyanosis and anorexia, initial hyperpnea, dyspnea and slow breathing, slow pulse, heart block, increased intracranial pressure with cerebral symptoms of confusion and moderate fever, paralysis and coma followed by clonic convulsions and possibly death due to circulatory collapse.
Methemoglobinemia: Case reports of clinically significant methemoglobinemia are rare at conventional doses of organic nitrates. The formation of methemoglobin is dose-related and in the case of genetic abnormalities of hemoglobin that favor methemoglobin formation, even conventional doses of organic nitrates could produce harmful concentrations of methemoglobin.
Treatment of Overdosage: Keep the patient recumbent in a shock position and comfortably warm. Passive movement of the extremities may aid venous return. Administer oxygen and artificial ventilation, if necessary. If methemoglobinemia is present, administration of methylene blue (1% solution), 1-2 mg per kilogram of body weight intravenously, may be required. If an excessive quantity of Nitroglycerin spray has been recently swallowed, gastric lavage may be of use.
Therapeutic Class
Nitrates: Coronary vasodilators
Storage Conditions
Store in a cool and dry place away from light and heat. Keep all medicines out of the reach of children.
- Type Spray
- Tag
- Morbi leo risus
- Porta ac consectetur ac
- Vestibulum at eros