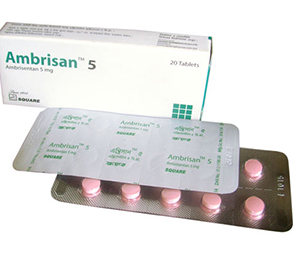
Ambrisan 5 1 Pc
Alternative products
Indications
Ambrisentan is
indicated for the treatment of pulmonary arterial hypertension (PAH)
(WHO Group 1):
- To improve exercise ability and delay clinical worsening.
- In combination with tadalafil to reduce the risks of disease progression and hospitalization for worsening PAH, and to improve exercise ability.
Studies establishing effectiveness included predominantly patients with WHO Functional Class II–III symptoms and etiologies of idiopathic or heritable PAH (60%) or PAH associated with connective tissue diseases (34%).
Therapeutic Class
Anti-hypertensive, Endothelin receptor antagonist
Pharmacology
Endothelin-1 (ET-1) is a potent autocrine and
paracrine peptide. Two receptor subtypes, ETA and ETB, mediate the effects of
ET-1 in the vascular smooth muscle and endothelium. The primary actions of ETA
are vasoconstriction and cell proliferation, while the predominant
actions of ETB are vasodilation, antiproliferation, and ET-1 clearance.
In patients with PAH, plasma ET-1 concentrations are increased as much as
10-fold and correlate with increased mean right atrial pressure and disease
severity. ET-1 and ET-1 mRNA concentrations are increased as much as 9-fold in
the lung tissue of patients with PAH, primarily in the endothelium of pulmonary
arteries. These findings suggest that ET-1 may play a critical role in
the pathogenesis and progression of PAH.
Ambrisentan is a high-affinity (Ki=0.011 nM) ETA
receptor antagonist with a high selectivity for the ETA versus ETB
receptor ( > 4000-fold). The clinical impact of high selectivity for ETA is
not known.
Dosage & Administration
Initial treatment is 5 mg once daily, and can be increased to 10 mg once daily if 5 mg is tolerated. Ambrisentan may be administered with or without food.
Interaction
Multiple dose co-administration of Ambrisentan and Cyclosporine resulted in an approximately 2-fold increase in Ambrisentan exposure in healthy volunteers; therefore, limit the dose of Ambrisentan to 5 mg once daily when co-administered with Cyclosporine.
Contraindications
Ambrisentan may cause fetal harm when administered to a pregnant woman. Ambrisentan is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to a fetus. Pregnancy must be excluded before the initiation of treatment with Ambrisentan and prevented during treatment and for one month after stopping treatment. Ambrisentan is contraindicated in patients with Idiopathic Pulmonary Fibrosis (IPF) including IPF patients with pulmonary hypertension (WHO Group 3).
Side Effects
Decreases in hemoglobin concentration and hematocrit have followed administration of other endothelin receptor antagonists and were observed in clinical studies with Ambrisentan.
Pregnancy & Lactation
Pregnancy Category X. It is not known whether Ambrisentan is excreted in human milk. Breastfeeding while receiving Ambrisentan is not recommended.
Precautions & Warnings
Fluid Retention: Peripheral edema is a known class effect of
endothelin receptor antagonists, and is also a clinical consequence of PAH and
worsening PAH.
Pulmonary
Veno-occlusive Disease:
If patients develop acute pulmonary edema during initiation of therapy with
vasodilating agents such as Ambrisentan, the possibility of pulmonary
veno-occlusive disease should be considered, and if con_rmed. Ambrisentan
should be discontinued.
Hematological
Changes: Decreases in
hemoglobin concentration and hematocrit have followed administration of other
endothelin receptor antagonists and were observed in clinical studies with
Ambrisentan.
Use in Special Populations
Pediatric patients: Safety and effectiveness of Ambrisentan
in pediatric patients have not been established.
Hepatic
impaired patient: Ambrisentan is
not recommended in patients with moderate or severe hepatic impairment.
Storage Conditions
Store in a cool and dry place, below 30° C. Protect from light and moisture.
- Type Tablet
- Tag
- Morbi leo risus
- Porta ac consectetur ac
- Vestibulum at eros