Lutisone Oint 1 Pc
Alternative products
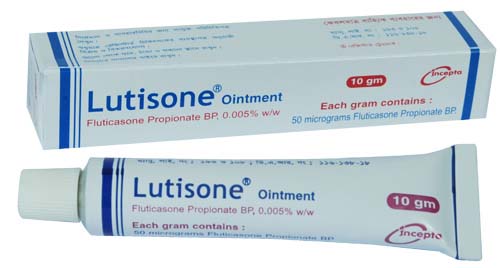
Fluticasone Propionate
Indications
Fluticasone Propionate is indicated for-
- The prophylaxis and treatment of seasonal allergic rhinitis including hay fever and perennial rhinitis.
- It has potent anti-inflammatory activity on the nasal mucosa without detectable systemic activity.
Description
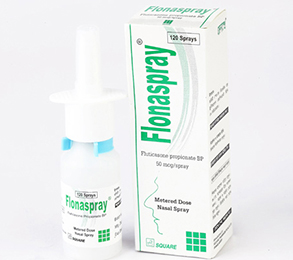
Fluticasone Propionate is a synthetic trifiuorinated corticosteroid with anti-inflammatory activity. It is an aqueous suspension of microfine Fluticasone Propionate for topical administration to the nasal mucosa by means of a metering, atomizing spray pump. Each bottle of Fluticasone Nasal Spray provides 120 metered sprays.
Pharmacology
Following topical administration into the nasal mucosa, Fluticasone propionate produces anti-inflammatory and vasoconstrictor effects. The exact mechanism of these actions remains unknown but may involve a reduction in the following: number of mediator cells (basophil, leukocytes and mast cells) at the epithelial level, number of eosinophils, the sensitivity of sensory nerves to mechanical stimuli, secretory response to cholinergic receptor stimulation, and fibroblast activity. Other mechanisms may involve - inhibition of capillary dilation and permeability, stabilization of lysosomal membranes and subsequent prevention of release of proteolytic enzymes.
Dosage
Adults & children over 12 years: 2 sprays in each nostril once a day preferably in the morning. In some cases 2 sprays into each nostril twice daily, not exceeding 4 sprays.
Children under 12 years (4-11 years): 1 spray in each nostril once a day. The maximum total daily dosage should not exceed 4 sprays. For perennial rhinitis in children, there are insufficient clinical data to recommend its use.
Children below 4 years: The safety and effectiveness of Fluticasone Propionate nasal spray in children below 4 years of age have not been established.
Patients should use Fluticasone Propionate Nasal Spray at regular intervals as directed since its effectiveness depends on its regular use. Or as directed by the physician.
* চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Administration
How to use the Nasal Spray-
- Shake the bottle gently and remove the dust cover.
- Hold the spray with your forefinger and middle finger on either side of the nozzle and your thumb underneath the bottle. Press down until a fine spray appears. If using for the first time or if you have not used it for a week or more, press the nasal applicator several times until a fine moist comes out from the container.
- Gently blow the nose to clear the nostrils.
- Close one nostril and carefully insert the nasal applicator into the open nostril. Tilt your head forward slightly and keep the spray upright. Breathe in through your nose and while breathing in, press the white-collar of nasal applicator firmly down once to release a spray.
- Breathe out through your mouth.
- Repeat the above steps in the same/ other nostril for consecutive doses.
Cleaning: The nasal spray should be cleaned at least once a week. The procedures are as follows-
- Remove the dust cover.
- Gently pull off the nasal applicator.
- Wash the applicator and dust cover in warm water.
- Shake off the excess water and leave to dry in a normal place. Avoid to apply additional heat.
- Gently push the applicator back on the top of the bottle and re-fix the dust cover.
* চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Contraindications
Fluticasone Propionate nasal spray is contraindicated in patients with a hypersensitivity to any of its ingredients.
Side Effects
Fluticasone Propionate nasal spray is absorbed less into the rest of the body, therefore fewer side effects are seen. With the nasal spray, drying of the nose and throat, unpleasant taste and smell and an increase in the incidence of nosebleeds may occur.
Pregnancy & Lactation
There are no adequate and well-controlled studies in pregnant women. Fluticasone Propionate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is not known whether Fluticasone Propionate is excreted in human breast milk. Because other corticosteroids are excreted in human milk, caution should be exercised when Fluticasone Propionate nasal spray is administered to a nursing woman.
Precautions & Warnings
Rare instances of wheezing, nasal septum perforation, cataracts, glaucoma and increased intraocular pressure have been reported following the intranasal application of corticosteroids, including Fluticasone Propionate. Although systemic effects have been minimal with recommended doses of Fluticasone Propionate nasal spray. Potential risk increases with larger doses. Therefore, larger than recommended doses of Fluticasone Propionate nasal spray should be avoided.
Therapeutic Class
Fluocinolone & Combined Preparations, Nasal Steroid Preparations
Storage Conditions
Keep all medicines out of reach of the children. Store in a cool and dry place protected from light.
- Type Ointment
- Tag
- Morbi leo risus
- Porta ac consectetur ac
- Vestibulum at eros